 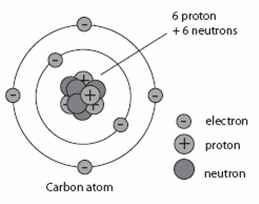  
Model, Lord Rutherfords Atomic model, Neils Bohrs Model of an Atom. The Bohr Model has an atom with a positively-charged nucleus surrounded by negatively-charged electrons that have circular, planetary-like orbits. In this model n = ∞ corresponds to the level where the energy holding the electron and the nucleus together is zero. Explain how the atomic number and mass number of carbon are 6 and 12 respectively. The Bohr Model is a modification of an earlier atomic model, the Rutherford Model. Bohr theory modified the atomic structure model by explaining that electrons move in fixed orbitals (shells) and not anywhere in between and he also explained. \) is the Rydberg constant in terms of energy, Z is the atom is the atomic number, and n is a positive integer corresponding to the number assigned to the orbit, with n = 1 corresponding to the orbit closest to the nucleus. 6 : 3034 ( J ) beta decay, effect of Bohr orbit binding on, 5 : 6366 ( J ) capture, evaporation theory of particle emission following, 7 : 1206 ( J ). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
